
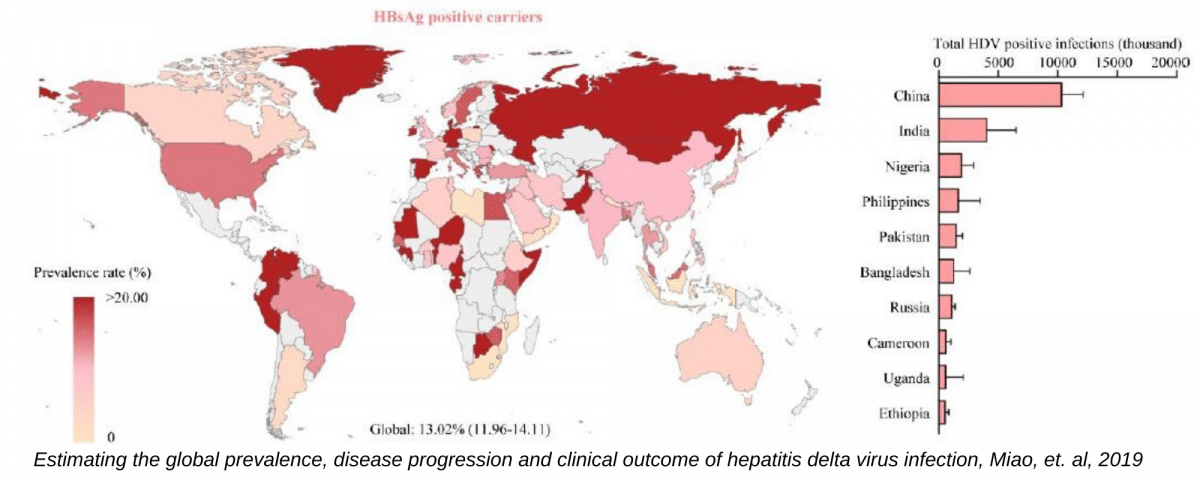
For decades, hepatitis delta, the dangerous coinfection of hepatitis B, was thought to only affect about 5-10% of the estimated 292 million people worldwide with chronic hepatitis B infections. With limited data and funding for research related to this complicated virus, true prevalence data, diagnostic tools and skilled physicians to manage hepatitis B and delta coinfection have remained limited until recent years. Publications in 2019 by Miao, et al., Chen, et al., Shen, et al., are helping to reveal a possibly more accurate picture of the burden of coinfection, conducting meta-analyses comprising data from hundreds of thousands of hepatitis B patients and the general population. While it was previously thought that 15-20 million coinfections existed globally, this new research has suggested there may be between 48-74 million1,2,3. Although these studies analyzed data that classified the presence of hepatitis delta antibodies, which can be present in cases of both past or current infection, there is a strong correlation between their presence and likelihood of an ongoing infection. These new studies may place coinfection at upwards of 10-15% of those with hepatitis B, with some of the hardest hit areas facing coinfection rates greater than 30%, in regions like Central Asia, Eastern Europe, Central Latin America and West and Central Africa1,2,3.
Understanding hepatitis delta is vital to helping to identify coinfected patients, who require altered treatment and management plans, and who may progress to cirrhosis and/or liver cancer in periods as little as 5-10 years. Diagnosis and management for hepatitis delta is still a challenge in much of the world, but in the US, it is becoming easier than ever before, with Quest Diagnostics, a commercial U.S. lab, rolling out a new HDV RNA test, a game-changer for physicians to easily order the test and manage patients. HDV RNA testing was previously available only through the Utah-based lab, ARUP, and Boston’s Cambridge Biomedical, but had to be specialty ordered. As testing continues to become more widely available and affordable, hepatitis B patients can more easily access testing. The more patients who are diagnosed, the more evidence for the urgent need for improved treatments to combat the virus, which is currently poorly controlled by the only available treatment; pegylated interferon.
Luckily, the virus has attracted the attention of nine pharmaceutical companies from around the world, with each working on a different approach to better controlling, or even curing hepatitis delta. Two of these companies, Eiger Biopharmaceuticals (US) and Myr Pharma (Germany) are now in Phase 3 clinical trials, where patients are flocking to enroll in these trials, which present new opportunities to receive treatments that may be more effective in controlling their coinfection. Eiger’s clinical trials will test their new drug, Lonafarnib, in clinical trial arms with and without pegylated interferon and/or ritonavir, with sites open in many countries throughout the world. Myr’s clinical trial will test their new drug, Myrcludex B, in similar triple-treatment combinations. Their clinical trial sites are now open in Russia, and the drug is already being prescribed for “compassionate use” in France. The United States Food and Drug Administration (FDA) has even taken notice; issuing guidance for industry on the development of hepatitis delta drugs for treatment in October 2019. This provides a valuable set of standards and expectations for clinical trials in regard to ethics, trial design, and patient needs.
Hepatitis delta coinfection has also received more attention this year at international hepatology conferences such as at the European Association for the Study of the Liver (EASL)’s International Liver Congress in Vienna, Austria, the International Liver Congress in American Association for the Study of the Liver (AASLD) meeting in Boston, and HEP DART in Hawaii. This year has brought many milestones for hepatitis delta data, diagnostics, and clinical trials. With continued scientific research and interest, Hepatitis Delta Connect hopes to continue to support these milestones and drive awareness efforts.
References:
1. Zhijiang Miao, Shaoshi Zhang, Xumin Ou, Shan Li, Zhongren Ma, Wenshi Wang, Maikel P Peppelenbosch, Jiaye Liu, Qiuwei Pan, Estimating the global prevalence, disease progression and clinical outcome of hepatitis delta virus infection, The Journal of Infectious Diseases, jiz633.
2. Chen H, Shen D, Ji D, et al. Prevalence and burden of hepatitis D virus infection in the global population: a systematic review and meta-analysis. Gut 2019;68:512-521.
3. Shen D, Ji D, Chen H, et al. Hepatitis D: not a rare disease anymore: global update for 2017–2018. Gut Published Online First: 09 April 2019.

